Cleanliness of space electronics hardware
Surface contamination involving micrometric particles, microbiological agents, molecular adsorbate and others, represent a significant hazard in many areas of industry. For instance, a high level cleanliness is a critical requirement that any EEE device has to fulfil at any development stage: from the fabrication process of single components, before encapsulation, to the final PCB assembly. This is so because micro- and nanometric foreign materials may produce corrosion, electrochemical migration, delamination or cause parametric changes. These and other contamination-induced phenomena are responsible for impairment performance, incorrect test results and, most importantly, the catastrophic fail of complete units. Beside such adverse effects, non-conducting contaminants can also contribute to electrostatic discharge, particularly in the space environment. Therefore this is a vital concern for different national and international aerospace, military and space agencies, which have adopted strict protocol about this issue. In particular the “Product Cleanliness Levels and Contamination Control Program” (IEST-STD-CC1246D) adopted by the USA “Defense and Logistics Agency” the “Process Specification for Cleaning of Hardware” (PRC-5001, Rev F) issued by the NASA and mainly the “Cleanliness and contamination control” and “Ultracleaning of flight hardware” (ECSS-Q-ST-70-01C and ECSS-Q-ST-70-54C respectively) developed by the ESA specifically state the acceptable cleanliness levels according to the targeted applications and components, as well as the preferred cleaning protocols.
Conventional cleaning methods
ECSS specifications thoroughly describe different recommended procedures such as blasting, mechanical, chemical, solvent, and thermal based cleaning methods. Nonetheless, they have to be carefully selected depending on the target contaminant, the chemical and physical nature of the contaminated item, and the ground phase. In particular, the ECSS-Q-ST-70-54C standard specifically warms about the potential detrimental effects of unsuitable cleaning procedures, particularly in the case of delicate components (e.g. those related to strong acids and bases, the use of organic solvents over plastic components, or thermal methods on temperature sensitive components).
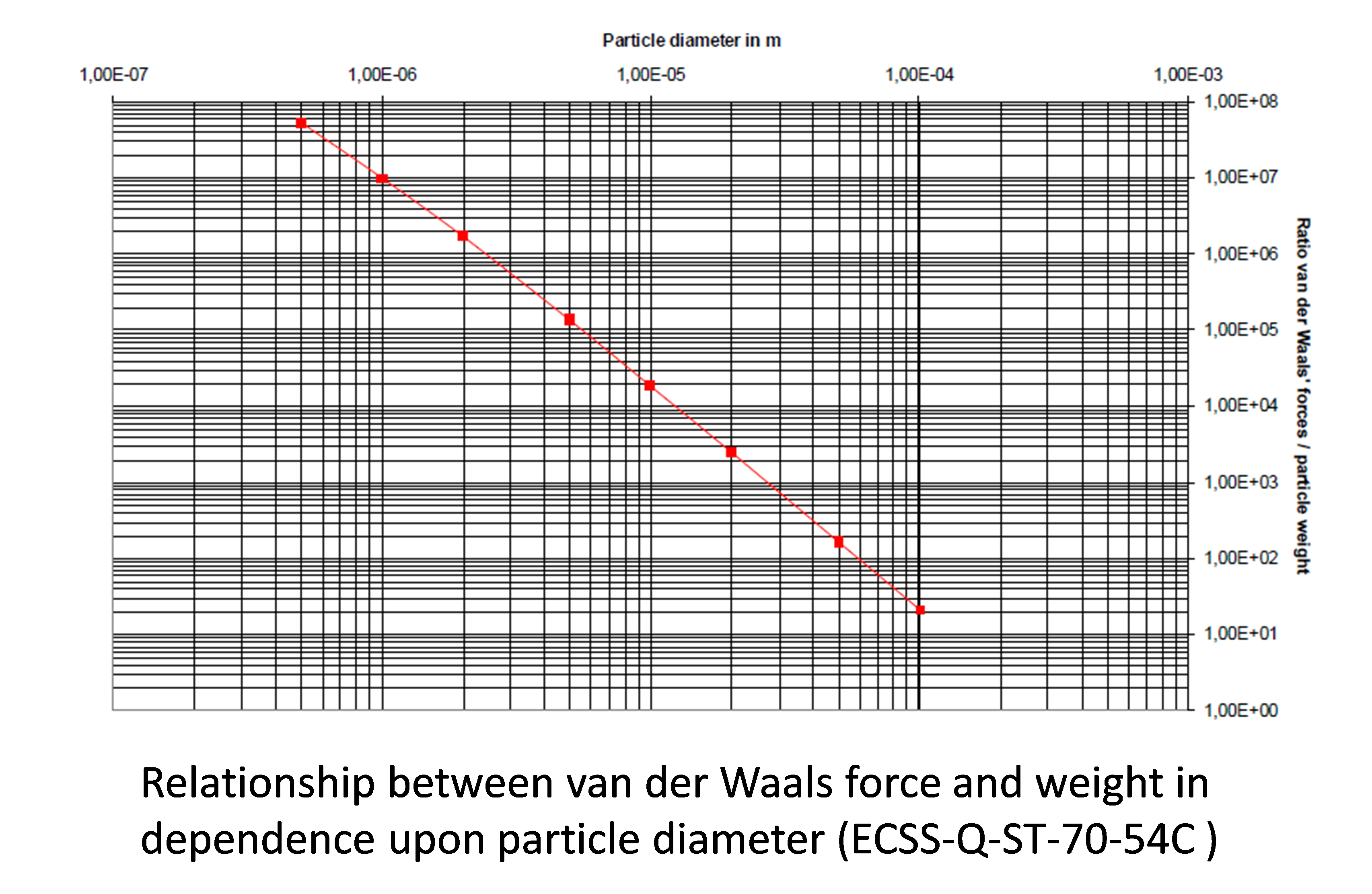
On the other hand, the suitable removal of micro and submicrometric contaminants is always a difficult task due to the very strong physicochemical interactions that they develop with the surface: covalent bonding, ionic bonding, van der Waals forces, hydrogen bonding, dipole-dipole and electrostatic interactions, or a combination of these. For instance, van der Waals forces linearly scales with the particle diameter whereas the weight decrease with the cubic diameter. Hence the van der Waals force to weight ratio sharply increases as the particle diameter is reduced and reach values of 108 for submicrometric systems, as it is illustrated in the figure. . All them are factors that increase the energy required for removal when considering pure physical methods and entail using stronger solvent, which in turn increase the risk of damage of the base materials. In addition, conventional solvents may also leave additional residues and be ineffective in the case complex-shaped components including micrometric cavities. In summary most of the approaches are not enough effective to reach the ultraclean levels demanded many space applications.
Why Plasma?
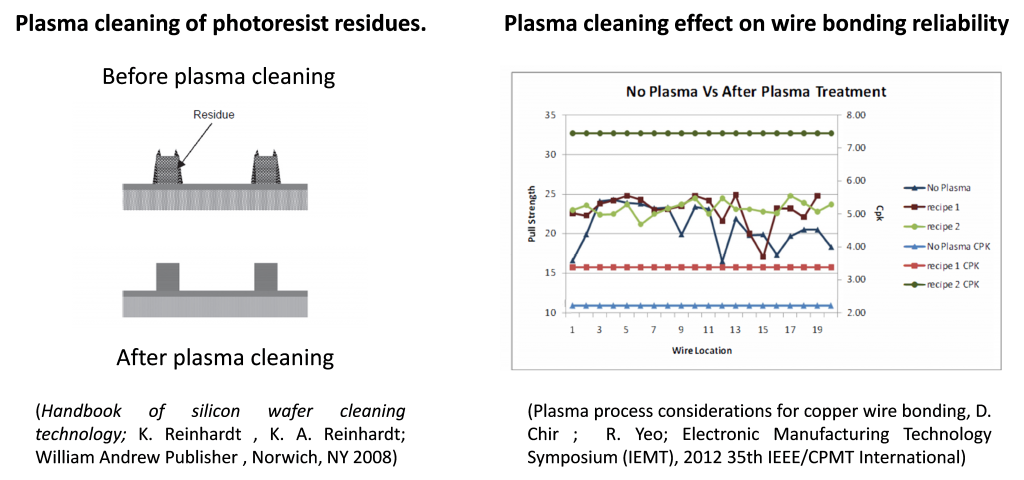
 A plasma discharge consists of a partially ionized gas at local level where the generated charged species exhibit a collective behaviour. From the point of view the high ionization and energetic level of the involved species it is considered the fourth state of the matter. Under specific conditions plasma discharges are characterized by the lack of thermal equilibrium between the “hot” free electrons and the rest of heavy species (atoms, ions, molecules, and radicals) defining the gas temperature. This makes it possible to induce highly energetic chemical reactions (e.g. 2CO2 → 2CO + O2 (135 kcal/mol) at room temperature, thanks to the interaction with the “hot” free electrons. Therefore, plasma processes are present in different fabrication steps of microelectronics components including the final fine removal of photoresist residues. On the other hand plasma treatment methods are also used to increase the surface wettability leading to an outstanding improvement of the underfilling during the flip-chip packing processes. Similarly plasma discharges are used for the activation of printed circuit boards prior to potting and encapsulation. Moreover, they are find applications for the de-oxidation of gold contact for improved wire boding.
A plasma discharge consists of a partially ionized gas at local level where the generated charged species exhibit a collective behaviour. From the point of view the high ionization and energetic level of the involved species it is considered the fourth state of the matter. Under specific conditions plasma discharges are characterized by the lack of thermal equilibrium between the “hot” free electrons and the rest of heavy species (atoms, ions, molecules, and radicals) defining the gas temperature. This makes it possible to induce highly energetic chemical reactions (e.g. 2CO2 → 2CO + O2 (135 kcal/mol) at room temperature, thanks to the interaction with the “hot” free electrons. Therefore, plasma processes are present in different fabrication steps of microelectronics components including the final fine removal of photoresist residues. On the other hand plasma treatment methods are also used to increase the surface wettability leading to an outstanding improvement of the underfilling during the flip-chip packing processes. Similarly plasma discharges are used for the activation of printed circuit boards prior to potting and encapsulation. Moreover, they are find applications for the de-oxidation of gold contact for improved wire boding.
Plasma cleaning
ESA specifications state plasma cleaning as an advantageous approach for the efficient removal of organic contaminant at ultraclean levels. Amongst other features it represents a very gentle, solventless and room temperature process for the removal of surface contaminants from delicate surfaces. Due to the high reactivity and energy of the generated species plasma processes may covers a variety of applications since the sputtering of inorganic surfaces to the soft plasma functionalization of organic materials. The plasma characteristics (e.g., electron energy distribution, ion energy distribution, residence time) that regular the delivery of energy and the intensity of the plasma interactions are controlled by the pressure, the plasma gases combination, and the injected power.
Due to the high reactivity oxygen is one of the preferred gases for plasma cleaning. For this gas the mechanisms involved in the plasma cleaning process are the following:
- Plasma excitation always involves the emission of ultraviolet photos at energies high enough to induce the fragmentation of the organic bounds of
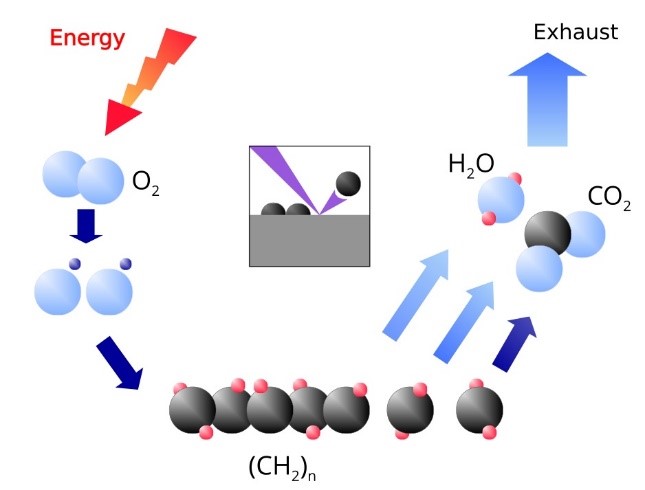 the contaminants (which are labile) without affect inorganic systems. Low energy ion bombardment induces a similar effect.
the contaminants (which are labile) without affect inorganic systems. Low energy ion bombardment induces a similar effect. - O2 molecules dosed into the plasma discharge dissociate and lead to generation of charged (e.g. O2–, O–, O2+, O+) and activated species. These activated species are molecules or atoms that in the very energetic plasma environment become promoted to a higher quantum state level and are very reactive towards the molecular fragments generated in the step 1.
- The reaction between the molecular fragments and the plasma species lead to formation of low molecular weight organic moieties with a rather high vapour pressure which are able of being pumped to the atmosphere.
Depending of the used base material and the targeted contamination other gases can be used. Thus hydrogen (H2), which is a powerful reducing agent is used to clean metals from oxide films. On the other hand, argon (Ar), which is not chemically reactive, generates heavier ions generated that exercise a micro-sandblast effect where they impinge on the surface. This cleans the surface without modifying the oxidation state. Argon plasma or in combination with other gases is able to remove even silicones and fibers.
Plasma cleaning processes are intended to react with the contamination layer without affecting the underlying film. This makes it necessary to select the appropriate gases combination, plasma conditions and the conception of a suitable plasma reactor configuration (for instance to avoid excessive ion bombardment). With this regards the plasma cleaning processes used at Alter Technology are carried out in a remote plasma reactor specifically conceived by Nanotechnology of Surfaces group of the Institute of Materials Science of Seville to finely regulate the plasma-substrate interaction which has proved his compatibility with delicate substrates.[DOI 10.1039/C4TC00294F].
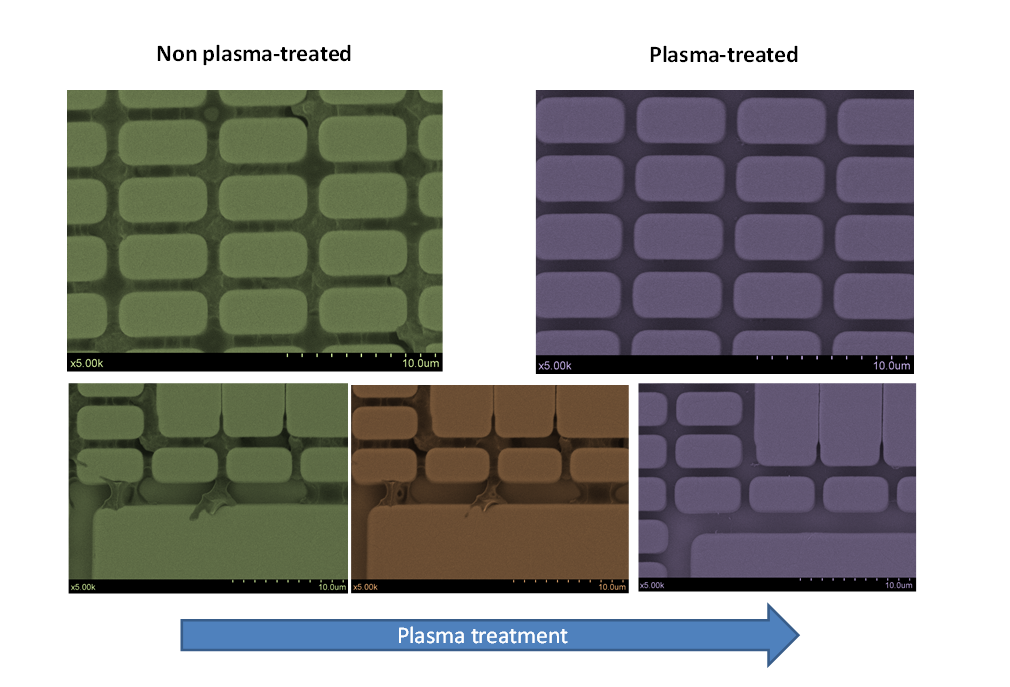
The image shows one example of the plasma cleaning performance when applied to a ASIC system that presented a large accumulation of unknown organic residues within the trenches. The images at the top correspond to a non-treated and a treated region, whereas the images at the bottom show the effect at different plasma treatment stages.
- Quantum Key Distribution - 7th November 2022
- Conducted Immunity - 20th May 2019
- Electrical transients Test - 2nd May 2019




