Corrosion/Contamination
Crimped connectors are gas-tight junctions ideally protected against environmental contaminants and corrosive agents. Nonetheless inappropriate crimping and inherent connector and wire defects reduce this protection. Thus, depending of the storage atmosphere, non-conducting corrosion products can be formed, or stress corrosion cracking can be eventually induced. The former act as insolation layer that reduces the connector conductivity and both corrosion effects are identified as critical factors involved in common fails reported for electrical connectors. Similarly, organic surface contamination at the contact interface forming and insulation or low resistance layer may also impair the system performance.
Creep corrosion is that related to platting and under plating defects that expose the base metal (Cu or Cu alloys) to oxygen or other corrosive agents. Typical Ni underplating blocks pore-initiated corrosion. However, when it reaches the base metal corrosion products may migrate towards the surface, which reduces the contact conductivity in a large extend as illustrated in the figure.
Galvanic corrosion is a corrosion process whereby the presence of an electrolyte (e.g. moisture) at the interface of two metals or alloys of dissimilar electrode potential promotes the accelerated corrosion of the one with more negative electrode potential (anode). In the case of crimped connectors such galvanic cells can be formed between the wires and the barrel (inadequate wire and barrel selection); or between the conductor base material and the corrosion protective coating (damaged plating). A typical example of a base-plaiting pair susceptible to galvanic corrosion are Cu and Ag which are responsible for the denominated “red plague”.
The figure shows, an example of Ag-Cu wire affected by red plague. Considering the load applied during crimping processes, Ag-Cu terminations are not recommended for crimped connectors unless additional measures are taken to prevent the red plague development (i.e. heavier plating thickness, environmentally rated connector, conformal coating, shrink sleeving, etc.)
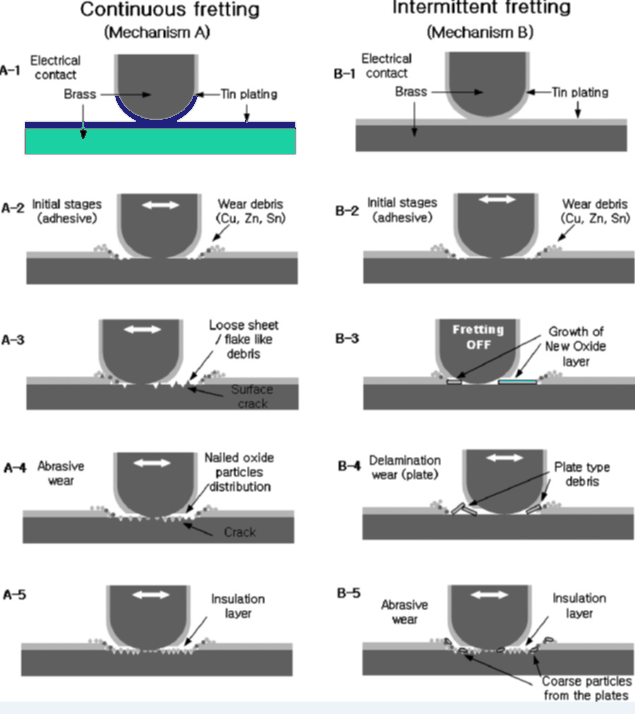
Fretting Corrosion, which is also called friction oxidation, refers to the corrosion mechanism accounted by the wear that the contact interface experience because of high frequency relative motion (vibration). This leads to the breakage of the contact interface. The resulting debris oxidize and are accumulated at the contact interface resulting in the formation of a thick insulating layer.
In the case of crimped connectors, fretting corrosion is related to those situations where the connector is slightly loose (for instance due to intense vibration). This is more likely for Cu based connectors due to the inherent high ductility.
Organic residues may be present at the contact interface due to several reasons including incorrect crimping and/or cleaning. In addition, they can be thermally released from surrounding plastic materials as the wire insulator. Such outgassing processes are induced by abnormal wire heating usually related to electrothermal events described in the next section.
Diagnosis tools: Corrosion products can be detected by different approaches being naked eye and external high-magnification optical inspection the quicker ones. Nevertheless, due to the close structure of crimped connectors microsection techniques are required to assess the presence of corrosion sings at the contact interface.
In addition, analytical techniques able to determine the oxidation state (e.g. x-ray photoemission spectroscopy) and microanalysis tools combing analytical and imaging capabilities (e.g. energy-dispersive X-ray spectroscopy) are essential for a successful failure analysis.
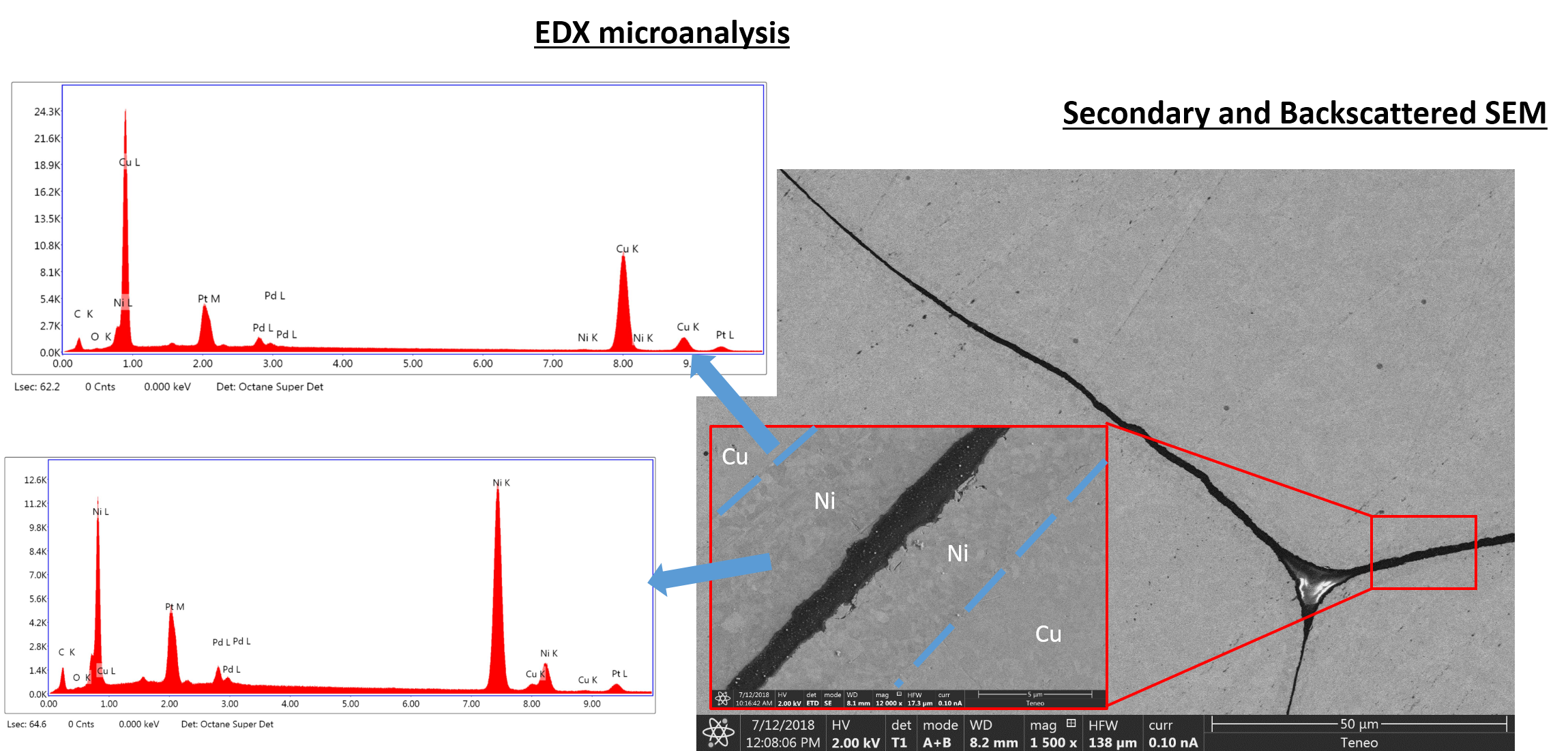
Example of electron microscopy microanalysis conducted by our laboratory.
Failures in crimped connectors can be can be related to different causes, some of the discussed elsewhere. The present post analyzes those related to corrosion and contamination phenomena.
Contact us
- Scanning Acoustic Microscopy on Ceramic Capacitors - 18th May 2020
- Non-destructive detection of micrometric internal features within EEE microelectronic systems. - 3rd September 2019
- Acoustic Inspection of Hybrid Systems on Laminated Substrates - 3rd September 2019